Immunotherapy Treatments
Immunotherapy Treatments
Immunotherapy is a biological therapy using substances produced from the body or in a laboratory. It is used to improve an individual’s immune system to fight diseases we would usually be defenceless against. It can be used for different purposes, reducing a cancer’s growth, inhibiting the cancer from spreading, or improving the patient’s immune system fight against the cancer itself. Immunotherapy is sensitive and very new to science and medicine, with most treatments still in trial stages. There are several types of immunotherapies out there all with a different approach to destroying or preventing cancer.
- Monoclonal antibodies (MAbs) are laboratory-produced antibodies designed to target protein expressions on cells
- Oncolytic virus therapies manipulate viruses to infiltrate and infect a tumour
- T-cells therapies take T-cells from a patient to genetically engineer them to be responsive to a desired antigen
- Cancer vaccines target infectious agents that cause or contribute to the development of cancer
- Non-specific cancer therapies boost the immune system in a more general way
Cancer
Cancer is infamous for its ability to avoid and escape the immune system. As cancer is caused by a genetic mutation within the cells native to the body, it can slip under the radar of the immune system, avoiding detection and destruction. This is due to the fact the immune system is designed to not target cells from its own body and if it did, it would cause many complications. Mutations in gene sequences occur readily, sometimes when a cell copies its DNA incorrectly. Mutations can also result from environmental factors, such as the types of food you eat, whether you smoke or how much sunlight your skin absorbs. Many of these mutations are harmless and do not usually result in cancer. A tumour suppressor protein, known as p53, checks to see if any DNA damage has occurred. It can temporarily stop the cell cycle and activate DNA repair, or if the damage is irreparable it can force the cell to self-destruct by initiating apoptosis (programmed cell death). If any of these failsafe pathways are disrupted, the cell can replicate rapidly and avoid programmed cell death, leading to tumour formation. If cancerous cells are located near blood vessels, they may detach from the main tumour and travel to another area of the body and proliferate there, spreading the cancer.
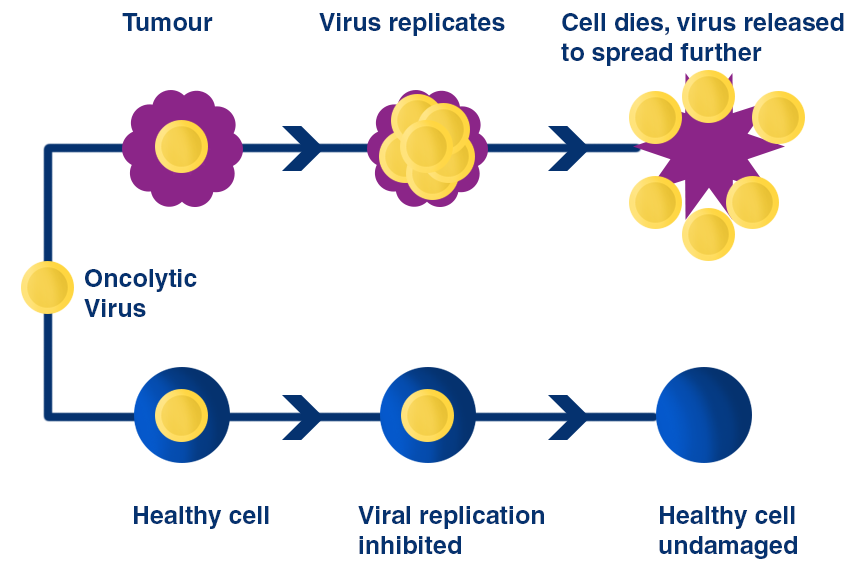
Oncolytic Virus Therapy
Oncolytic virus therapy introduces a new type of therapy for cancer patients. An oncolytic virus is a virus that is engineered to only target cancer cells, leaving healthy cells intact. It has been observed to infiltrate and destroy cancer cells directly, while also generating systemic anti-tumour immunity. The first oncolytic virus to be clinically approved for melanoma is talimogene laherparepvec (T-VEC), a type 1 herpes simplex virus designed to replicate inside and kill a tumour cell. T-VEC has been modified to include the gene that codes for granulocyte-macrophage colony-stimulating factor (GM-CSF), a protein responsible for the production of immune cells. As it replicates, it bursts the cancer cell, destroying it. When the cell dies it releases GM-CSF, other viruses and a variety of tumour-specific antigens, stimulating an immune response throughout the body. Injected and un-injected tumours both had a durable response rate, indicating that this therapy is capable of generating a systemic immune response. Full understanding of exactly how it achieves this is still unknown.
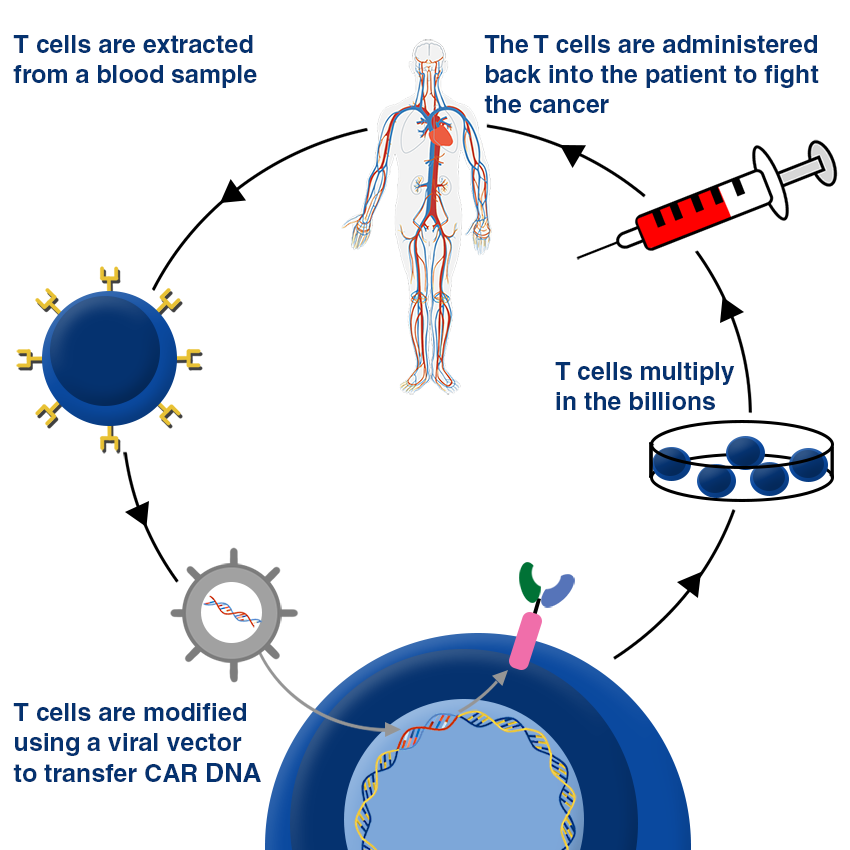
CAR T cell Therapy
Chimeric Antigen Receptor T-cell (CAR T-cell) therapy uses the body’s natural T-cells to combat cancerous growths. T-cells are extracted from the individual and taken to a specialised laboratory to be modified to detect antigens present on the cancer cells. This process involves modifying an antibody, designing it for the desired target, and then fusing it together with the extracted T-cell. The T-cell can now recognise an antigen on the target tumour with its new infused receptors present on its surface, a capability it could never have before. The CAR T-cells are then cultured in the laboratory to multiply in the billions. Once re-introduced into the patient, CAR T-cells can seek out the antigens present on the tumours, destroying them. CAR T-cells are not drugs but are alive cells: they can replicate on their own and persist inside the body after treatment, therefore safeguarding the body against cancer recurrence.
Cancer Vaccines
Vaccines are an additional approach towards decreasing the number of people diagnosed with cancer. They are split into two separate categories, preventative and treatment. Preventive vaccines are intended to prevent the disease from occurring in the first place by presenting the immune system with a weakened variant of an infectious agent known to cause or contribute to cancer. T-cells recognise and destroy it and memory B-cells are developed, arming the immune system with a blueprint of the agent, in a similar fashion to how traditional vaccines work. The most known preventative cancer vaccine is for HPV, a virus known for its ability to cause abnormal growths in the body, usually causing some form for cancer.
On the other hand, treatment vaccines focus on improving the immune system for those already diagnosed with cancer. The aims of the treatment are to delay or stop growth, to prevent reoccurrence of cancer, and to provide an alternative treatment for those whose previous treatments were ineffective. Cancer cells can often have cancer-specific antigens present on their surface that the immune system is able to detect as foreign, but they may be in such small quantities that they are ignored. Treatment vaccines work by increasing the immune system’s ability to recognise these antigens as a threat to the body. Patients are given an injection of the antigens, stimulating antibody production and T-cell activation in the hope that the immune system will now recognise and eliminate cancer cells presenting the antigen. This treatment is usually given with adjuvant, a substance which improves the efficiency and speed of response to a foreign body.
Non-specific Cancer Therapy
Adjuvants are also used in non-specific cancer therapy, an approach that affects the body in a general way, boosting the immune response against cancer. They can be used as standalone treatments, or with other immunotherapies to improve the treatment’s effectiveness. Cytokines, which are a type of adjuvant, are small proteins with an important role of cell signalling. Interleukin-2 is a one of the many variants of Interleukin which are a group of cytokines. Specifically, interleukins-2 stimulates immune cell growth and division more rapidly than without. Interferons are also used in non-specific cancer therapy, the other contender from the cytokines group. IFN-alfa, one of the three types of interferons, plays a crucial role in supporting the immune system, boosting the ability of certain immune cells to attack cancer cells, as well as slowing the growth of cancer.
Monoclonal Antibody Therapy
Monoclonal antibodies (MAbs) are a useful resource in the medical field. MAbs are highly specific and bind with high affinity to a wide variety of different molecules. They have the ability to target specific types of cells and mediate tumour suppression which demonstrates greater efficacy over standard chemotherapy regimens which are much less targeted. Antibodies only bind to the epitope they were designed to bind, so healthy cells are not affected. MAbs are produced by hybridoma cells, the fusion of a mouse spleen cell with cells from a mouse myeloma cell. The spleen cell secretes the desired antibody whilst the myeloma cell allows the cell line to proliferate indefinitely and produce limitless supplies.
Ipilimumab is one of the many MAbs used to treat cancer, but is specifically for late stage melanoma that has spread deep into the skin where it cannot be removed by surgery, or has spread to other parts of the body. Ipilimumab is designed to target CTLA-4, a protein found on the surface of T-cells. This protein functions to hinder the T-cell from endogenous cells and causing an autoimmune response. Therefore CTLA-4 prevents T-cells from attacking cancer cells. Ipilimumab works by down regulating the effectiveness of the T-cell receptors, allowing them to recognize melanoma cells as a threat and work to eliminate them.
Inhibiting protein expressions on either T cells or tumour cells is not all MAbs are capable of. Their Fc receptors located on the bottom of their Y shape structure can have a variety of substances attached to it. Radioimmunoconjugates, are monoclonal antibodies chemically linked to a radionuclide. They can be used to provide specifically targeted radiotherapy, attaching to the antigens expressed on the tumour before unleashing a deadly dose of radiation. Similarly, monoclonal antibodies can be labelled with a chemotherapeutic drug, delivering the treatment directly to the cancer cells while avoiding healthy cells, something that’s usually difficult to avoid in standard chemotherapy treatments.
In Summary
Traditionally, we have treated cancer with either chemotherapy or radiotherapy, which has so far been our only defence against it. These therapies have improved in the years regarding their effectiveness through research and development but still come at a heavy cost of side effects and low survival rates. Chemotherapy is a blanket term for drugs that shrink, slow, or destroy rapidly dividing cells, a characteristic of tumours, but other healthy cells in the body, especially cells with a natural rapid cell cycle such as hair cells, are also harmed by this approach. Radiation therapy aims to destroy cancer cells by delivering high doses of radiation directly on to the tumour, causing the DNA in the cells to break apart. When this happens the cells can no longer grow or divide, causing them to eventually die. Healthy cells near the tumour are also affected by the radiation, but are more likely to recover from it. Despite being the only treatments to fight cancer for decades, traditional chemotherapy and radiotherapy are now a very outdated way to deal with cancer. Immunotherapies are targeted, advanced and up-to-pace with science and technology, producing significantly fewer side effects than both chemo and radiotherapy. Although there are currently many limitations, there is also great potential for development into a successful treatment. Immunotherapy is still in the extremely early stages for development, yet has already demonstrated a lot of promise by harnessing the power of our own bodies.
