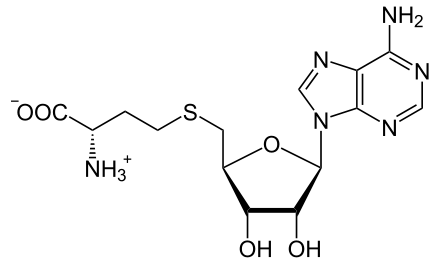
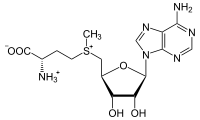
SAM and SAH
SAM and SAH
SAM was first discovered in Italy by Giulio Cantoni in 1952. Is a common cosubstrate involve in methyl group transfers, transsulfuration and aminopropylation.
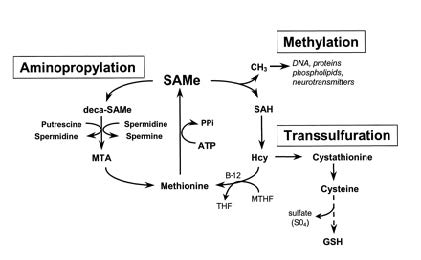
SAM cycle
The reactions that produce, consume, and regenerate SAM are called the SAM cycle. In the first step of this cycle, the SAM-dependent methylases (EC 2.1.1) that use SAM as a substrate produce S-adenosyl homocysteine as a product. This is hydrolysed to homocysteine and adenosine by S-adenosylhomocysteine hydrolase EC 3.3.1.1 and the homocysteine recycled back to methionine through transfer of a methyl group from 5-methyltetrahydrofolate, by one of the two classes of methionine synthases or cobalamin-independent. This methionine can then be converted back to SAM, completing the cycle. In the rate-limiting step of the SAM cycle, MTHFR (methylenetetrahydrofolate reductase) irreversibly reduces 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate.
S-adenosylmethionine (SAM) is the key molecule in methionine cycle. It plays essential roles in the synthesis and metabolism of nucleic acids, proteins as well as maintaining the mobility of cytoplasmic membranes.
S-Adenosyl-L-homocysteine (SAH) is an amino acid derivative used in several metabolic pathways in most organisms. SAH is formed by the demethylation of S-adenosyl-L-methionine (SAM).
SAH SAM